| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Anatomy and Physiology of the Bone Marrow
Related Subjects: |AP of the Oesophagus |AP of the Diaphragm |AP of the Large Bowel (Colon, Rectum, Anal Canal) |AP of Small Bowel |AP of the Biliary system |AP of the Bone Marrow |AP of the Eye |AP of the Pharynx |AP of the Larynx |AP of the Ear |AP of the Nose |AP of Male Genitalia |AP of the Breast |AP of the Stomach |AP of the Rectum |AP of the Spleen
Bone marrow is a highly vascular, soft connective tissue housed within the medullary cavities of long bones and the trabecular spaces of cancellous bone. It is the primary site of haematopoiesis from late fetal life onward, producing red cells, white cells, and platelets under tight hormonal and cytokine control. Two functional forms exist: red marrow (active blood formation) and yellow marrow (fat-rich reserve with a capacity to reconvert when demand rises). Bone marrow is also an immune organ, a stem cell niche, and a key microenvironment for malignant haematological disease.
🧬 Development & Distribution Across the Lifespan
- Fetal haematopoiesis (site shifts):
- Yolk sac (early embryonic) → liver (major fetal site) ± spleen → bone marrow predominates by ~5th month gestation.
- Neonatal pattern: most marrow is red (high haematopoietic demand).
- Age-related conversion: progressive replacement of red marrow by yellow marrow, starting in distal long bones and moving proximally.
- In healthy adults, red marrow is concentrated in the axial skeleton and proximal long bones.
- Reconversion (demand response): severe anaemia, haemorrhage, chronic hypoxia (e.g., advanced lung disease), or increased haematopoietic drive can cause yellow → red reconversion (radiology relevance on MRI).
🔎 Types of Bone Marrow
-
🟥 Red marrow (haematopoietic): produces RBCs, WBCs, platelets.
– Typical adult sites: pelvis (iliac crests), vertebrae, ribs, sternum, skull and proximal femur/humerus.
– Highly cellular; supported by stromal niche + rich sinusoidal blood supply. -
🟨 Yellow marrow (fat-rich): energy reserve and stromal support tissue.
– Typical adult sites: diaphyses (shafts) of long bones.
– Not “inactive”: adipocytes and stromal elements actively influence the niche, and it can revert to red when required.
🏗️ Bone Marrow Microanatomy (The “Niche”)
Marrow is best understood as a 3D ecosystem that regulates stem cell fate. The “niche” maintains a balance between quiescence (stem cell preservation), proliferation (response to demand), and differentiation (lineage output). Disruption of this environment contributes to marrow failure, fibrosis, and malignant infiltration.
- Extravascular haematopoietic cords: spaces between vessels where most maturation occurs.
- Sinusoids: thin-walled, fenestrated vessels lined by specialised endothelium:
- Act as a selective gate for mature cell egress into blood.
- Permit passage of deformable mature RBCs and platelets; immature cells are usually retained.
- Endosteal niche: near the bone surface (endosteum):
- Osteoblast-lineage and bone-lining cells help maintain HSC quiescence and long-term self-renewal.
- Perivascular niche: around sinusoids and arterioles:
- Key site for HSC trafficking and activation; includes pericytes and stromal cells that provide growth signals.
- Stroma (support scaffold): reticular stromal cells, fibroblasts, macrophages, endothelial cells, adipocytes, and extracellular matrix:
- Provides adhesion signals, chemokines, and cytokines that steer lineage decisions.
- Macrophages support “erythroblastic islands” (clusters of developing erythroblasts around a central macrophage that facilitates iron supply and enucleation).
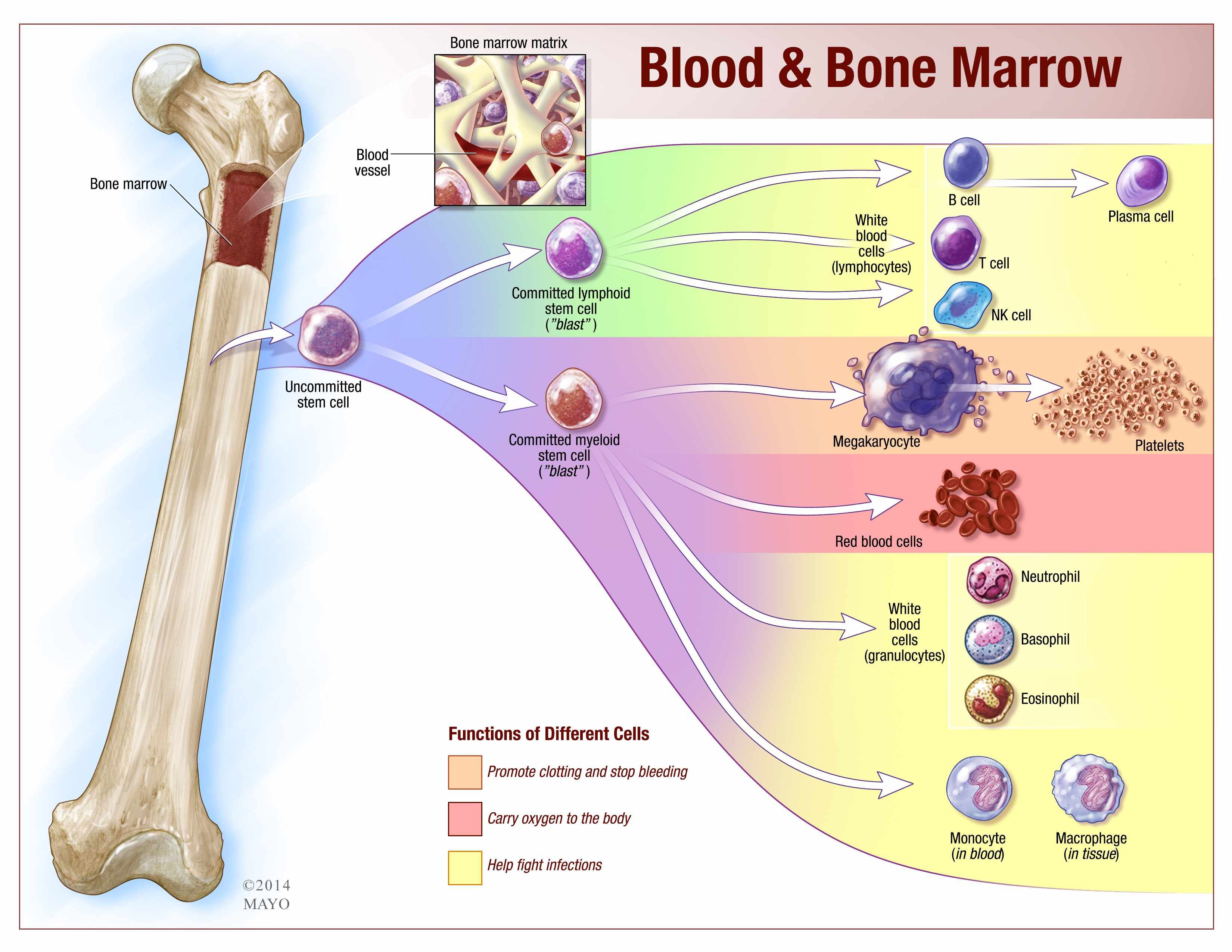
🩸 Haematopoiesis (How blood cells are made)
Haematopoietic stem cells (HSCs) are multipotent, self-renewing cells that generate all blood lineages. They first produce committed progenitors, then lineage-specific precursors that mature within the marrow cords. Mature cells then traverse the sinusoidal endothelium into the bloodstream-controlled by adhesion molecules and chemokine gradients.
🌱 Lineage Pathways
- Myeloid lineage:
- Erythrocytes (RBCs): oxygen transport; lifespan ~120 days.
- Megakaryocytes → platelets: haemostasis; platelet lifespan ~7–10 days.
- Granulocytes: neutrophils/eosinophils/basophils (innate immunity; neutrophils short circulating half-life).
- Monocytes → macrophages: phagocytosis, antigen presentation, tissue repair.
- Lymphoid lineage:
- B cells: mature in marrow; antibody production after activation (humoral immunity).
- T cells: progenitors originate in marrow but mature in thymus (cell-mediated immunity).
- NK cells: innate cytotoxic lymphocytes targeting virally infected and malignant cells.
⚙️ Physiology: Regulation of Blood Cell Output
Marrow output is matched to physiological demand: hypoxia drives erythropoiesis, inflammation and infection drive granulopoiesis, and platelet production adjusts to consumption. This is controlled by endocrine hormones, local cytokines, and marrow niche signals.
- Erythropoietin (EPO): mainly kidney-derived (peritubular interstitial fibroblast-like cells); rises in hypoxia → ↑ erythroid progenitors and RBC output.
- Thrombopoietin (TPO): mainly liver (also kidney); regulates megakaryocyte proliferation and platelet production.
- G-CSF / GM-CSF: stimulate granulocyte and myeloid progenitor proliferation and maturation.
📌 Used clinically after chemotherapy and for stem cell mobilisation into peripheral blood. - Interleukins:
- IL-3: multipotent progenitor support.
- IL-5: eosinophil lineage support.
- IL-7: lymphoid development (especially B/T lineage support).
- Iron, B12, folate: essential substrates for effective erythropoiesis (deficiency → ineffective or reduced RBC production).
- Inflammation (hepcidin): IL-6-driven hepcidin increases iron sequestration and reduces gut absorption → contributes to anaemia of chronic disease.
♻️ Extra Functions (Often Forgotten)
- Iron handling & recycling support: marrow macrophages recycle iron from senescent RBCs and provide iron to developing erythroblasts.
- Immune education: B-cell development and central tolerance checkpoints occur in marrow; antigen-presenting cells contribute to immune regulation.
- Stem cell reservoir: HSCs can be mobilised into blood (stress, G-CSF), and can home back to marrow via chemokine signalling (e.g., CXCR4–CXCL12 axis).
- Metabolic signalling: marrow adipocytes and stromal cells influence haematopoiesis through adipokines and cytokines (relevant in ageing and chronic disease).
🧪 Clinical Sampling (Aspirate vs Trephine)
- Common site: posterior iliac crest (safe access, rich red marrow).
- Aspirate: evaluates cell morphology, differential counts, flow cytometry, cytogenetics/molecular testing.
- Trephine (core biopsy): evaluates architecture, cellularity, fibrosis, infiltration (e.g., lymphoma, myelofibrosis, metastatic cancer).
- “Dry tap”: can occur in myelofibrosis or heavy infiltration (aspirate yields little), making trephine crucial.
⚠️ Clinical Relevance
- 🎗️ Leukaemia: malignant proliferation replaces normal marrow → anaemia, infection (neutropenia/dysfunction), bleeding (thrombocytopenia).
- 🚫 Aplastic anaemia: marrow failure with hypocellularity → pancytopenia (fatty replacement).
- 🩹 Myelofibrosis: marrow scarring → cytopenias + extramedullary haematopoiesis (splenomegaly); “dry tap”.
- 🧬 Myelodysplastic syndromes (MDS): ineffective haematopoiesis → cytopenias despite cellular marrow; dysplasia on morphology.
- 🧫 Infection/inflammation: marrow can show reactive changes (left shift, increased plasma cells) depending on stimulus.
- 🔄 Stem cell transplant: restores haematopoiesis after conditioning; graft-versus-leukaemia effect is a key mechanism in some malignancies.
📊 Quick Revision Table
| Feature | Red Marrow | Yellow Marrow |
|---|---|---|
| Main role | Haematopoiesis (RBCs, WBCs, platelets) | Fat storage + supportive stromal/niche functions |
| Typical adult location | Axial skeleton + proximal femur/humerus | Diaphyses of long bones |
| Dominant cells | HSCs, progenitors, maturing blood cells, stromal support | Adipocytes + stromal elements |
| Age trend | High in childhood → declines with age | Increases with age |
| Plasticity | Continuous output; rapidly upregulated with demand | Can reconvert → red in severe anaemia/bleeding/hypoxia |
Categories
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Critical care
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Geriatrics
- Guidelines
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Palliative Care
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Respiratory
- Resuscitation
- Revision Topics
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
