Related Subjects:
|Metabolic acidosis
|Anion Gap and Metabolic Acidosis
|Diabetic Ketoacidosis (DKA) Adults
|Alcoholic Ketoacidosis
|Euglycaemic Ketoacidosis (euDKA) with SGLT2 Inhibitors
|Causes of Ketoacidosis
⚠️ An elevated anion gap should be treated as potentially life-threatening until proven otherwise.
It usually reflects the presence of an unmeasured acid such as lactate, ketones, or toxins such as methanol or ethylene glycol.
It may also occur in uraemia. Always interpret the anion gap alongside the pH, bicarbonate, lactate, ketones, glucose, and the clinical picture.
📖 Definition and Normal Values
| Formula |
Expression |
Typical normal range |
Notes |
| Potassium excluded |
AG = Na+ − (Cl− + HCO3−) |
~8–12 mmol/L |
Most commonly used in clinical practice |
| Potassium included |
AG = (Na+ + K+) − (Cl− + HCO3−) |
~10–18 mmol/L |
Sometimes used in exams and older references |
🧠 Why the Anion Gap Rises
Plasma must remain electrically neutral. When acids accumulate, hydrogen ions are buffered by bicarbonate, so bicarbonate falls. The accompanying anions - such as lactate, beta-hydroxybutyrate, formate, or glycolate - are not routinely measured, so the anion gap increases. A raised anion gap therefore suggests an unmeasured acid.
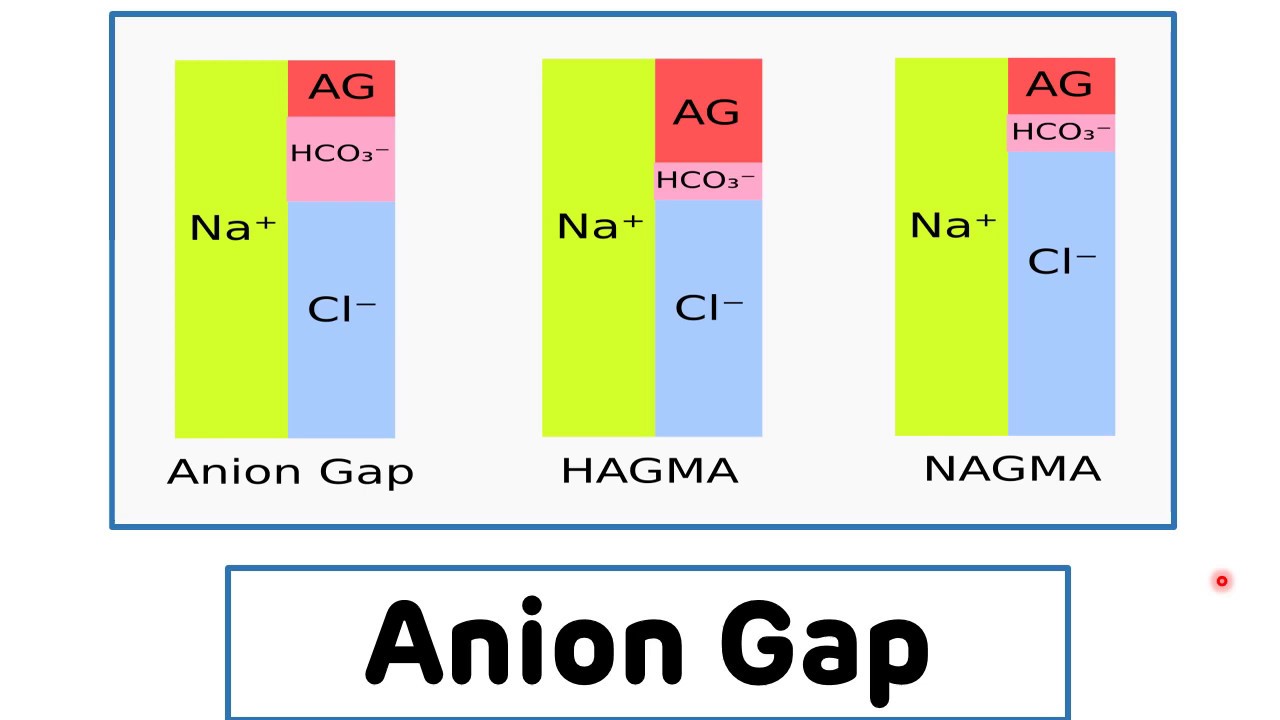
📊 High vs Normal Anion Gap Metabolic Acidosis
| Feature |
HAGMA (High anion gap) |
NAGMA (Normal anion gap) |
| Definition |
MA + raised AG |
MA + normal AG |
| Main mechanism |
Accumulation of unmeasured acids |
Bicarbonate loss or impaired renal acid handling |
| Chloride |
Usually normal |
Usually raised → hyperchloraemic acidosis |
| Typical causes |
Lactate, ketones, renal failure, toxins |
Diarrhoea, RTA, saline excess, acetazolamide |
| Clinical urgency |
Often immediately dangerous |
Can still be serious, but often less toxin-driven |
🩸 Causes of HAG Metabolic Acidosis (HAGMA)
| Category |
Examples |
Key clue |
| Ketones |
DKA, alcoholic ketoacidosis, starvation ketoacidosis |
Ketonaemia, diabetes, fasting, alcohol history |
| Lactate |
Sepsis, shock, hypoxia, mesenteric ischaemia, cyanide, CO, metformin |
High lactate, tissue hypoperfusion or toxin exposure |
| Renal failure |
Uraemic acidosis |
Raised urea/creatinine, CKD or AKI |
| Toxins / drugs |
Methanol, ethylene glycol, salicylates, iron, isoniazid, paraldehyde |
Poisoning history, osmolar gap, neuro/visual features |
💡 GOLD MARK Mnemonic
| Letter |
Meaning |
| G | Glycols (ethylene glycol, propylene glycol) |
| O | Oxoproline (5-oxoproline) |
| L | L-lactate |
| D | D-lactate |
| M | Methanol |
| A | Aspirin (salicylates) |
| R | Renal failure |
| K | Ketones |
🧂 Causes of Normal Anion Gap Metabolic Acidosis (NAGMA)
| Category |
Examples |
Mechanism |
| GI bicarbonate loss |
Diarrhoea, small bowel fistula, pancreatic drainage |
Direct bicarbonate loss |
| Renal causes |
Renal tubular acidosis, hypoaldosteronism, Addison’s, ureteroenterostomy |
Impaired acid excretion or bicarbonate handling |
| Drugs |
Acetazolamide, ammonium chloride |
Carbonic anhydrase inhibition or acid load |
| Iatrogenic |
Large-volume 0.9% saline infusion |
Hyperchloraemia lowers bicarbonate |
⚠️ Important Caveat: Albumin
| Issue |
Why it matters |
| Low albumin |
Albumin is a major unmeasured anion, so hypoalbuminaemia can make the anion gap look falsely normal |
| Clinical implication |
A “normal” anion gap does not reliably exclude dangerous acids in sepsis, liver disease, nephrotic syndrome, or malnutrition |
🔎 Practical Interpretation
| Step |
What to do |
Why |
| 1 |
Confirm metabolic acidosis |
Look for low pH with low bicarbonate |
| 2 |
Calculate anion gap |
Separates HAGMA from NAGMA |
| 3 |
If gap is raised, check lactate, ketones, renal function, toxins |
These are the major dangerous causes |
| 4 |
If gap is normal, think bicarbonate loss or RTA |
Chloride usually rises to maintain electroneutrality |
| 5 |
Check albumin and clinical context |
A normal AG can be misleading |
🖼️ Diagram
🩺 Clinical Cases
| Case |
Blood gas / labs |
Diagnosis |
Teaching point |
🩸 Case 1
70-year-old diabetic, drowsy, Kussmaul breathing, fruity odour |
pH 7.12, HCO₃⁻ 10, AG 26, glucose 28, ketones +++ |
DKA |
High AG due to ketoacid accumulation |
☣️ Case 2
45-year-old confused, tachypnoeic, hypotensive after overdose |
pH 7.19, HCO₃⁻ 9, AG 28, lactate high |
Lactic acidosis |
Commonest hospital HAGMA; think shock, sepsis, toxins, ischaemia |
🚽 Case 3
60-year-old with prolonged diarrhoea |
pH 7.28, HCO₃⁻ 15, AG 10, lactate normal |
NAGMA |
Bicarbonate loss with compensatory hyperchloraemia |
🌟 Summary Table
| If you see… |
Think… |
| Raised AG |
Unmeasured acid: lactate, ketones, renal failure, toxins |
| Normal AG + low bicarbonate |
Bicarbonate loss or renal tubular dysfunction |
| Normal AG but low albumin |
Possible hidden HAGMA |
📚 References

