| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Anatomy and Physiology of the Kidneys
Related Subjects: |Metabolic acidosis |Lactic acidosis |Acute Kidney Injury (AKI) / Acute Renal Failure |AP of the Kidneys |Chronic Kidney Disease (CKD) |Anaemia in Chronic Kidney Disease |Analgesic Nephropathy |Medullary Sponge kidney |IgA Nephropathy (Berger's disease) |HIV associated nephropathy (HIVAN)
The kidneys are paired, retroperitoneal organs (approximately T12–L3; left often slightly higher) that maintain homeostasis by regulating extracellular volume, electrolytes, acid–base balance, and blood pressure, while excreting metabolic waste and drugs. They receive a disproportionately high blood flow (~20–25% of cardiac output), making them excellent “filters” but also vulnerable to hypoperfusion and nephrotoxins (important in AKI pathways).
🩻 Gross Anatomy
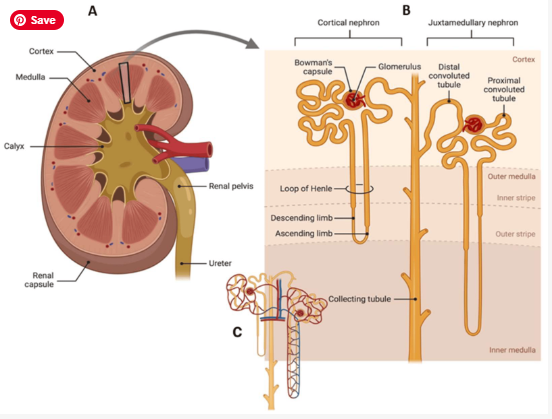
- Location: Retroperitoneal, either side of spine; protected by lower ribs and posterior abdominal wall muscles.
- Coverings: Fibrous capsule → perinephric fat → renal fascia (Gerota’s) → paranephric fat.
- Hilum: Medial indentation where renal vein, renal artery, lymphatics, nerves, and renal pelvis/ureter pass.
- Remember: Vein (anterior) → Artery → Pelvis/ureter (posterior) (V-A-P).
- Cortex: Glomeruli + proximal & distal tubules; high blood flow; highly metabolic.
- Medulla: Pyramids containing loops of Henle and collecting ducts; relatively low oxygen tension → prone to hypoxic injury.
- Papilla: Tip of pyramid draining into minor calyx → major calyx → renal pelvis → ureter.
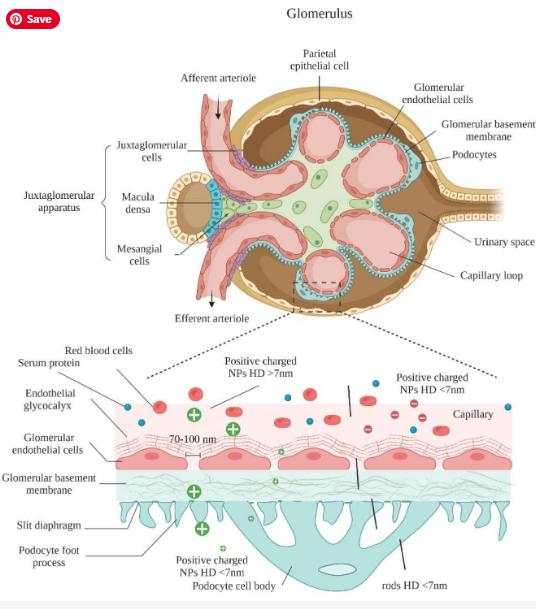
🧬 Microanatomy: The Nephron (Functional Unit)
Each kidney contains ~1–1.5 million nephrons. Renal function is the sum of filtration at the glomerulus plus selective tubular reabsorption and secretion. Nephrons are broadly cortical (most; short loops) and juxtamedullary (fewer; long loops essential for urine concentration).
| Segment 🧩 | Key transport & physiology ⚙️ | High-yield clinical links 🩺 |
|---|---|---|
| Glomerulus + Bowman’s capsule (renal corpuscle) |
|
|
| Proximal tubule (PCT) 🧽 |
|
|
| Loop of Henle 🔁 |
|
|
| Distal convoluted tubule (DCT) 🎛️ |
|
|
| Collecting duct 🚰 |
|
|
🫀 Renal Blood Supply & Microcirculation
Renal perfusion is high to support filtration, but intrarenal oxygenation is uneven: the cortex is well perfused, while the medulla operates at low oxygen tension. This is why hypoperfusion, sepsis, and vasoconstrictors can precipitate AKI, and why the medulla is particularly vulnerable.
- Arterial tree: Renal artery → segmental → interlobar → arcuate → interlobular (cortical radial) → afferent arterioles.
- Afferent arteriole → glomerulus → efferent arteriole (unique “two arterioles in series” arrangement).
- Efferent arteriole branches:
- Peritubular capillaries (cortex): support reabsorption/secretion.
- Vasa recta (medulla): countercurrent exchange preserves medullary gradient.
🧠 Renal Innervation & Hormonal Control
- Sympathetic (dominant): vasoconstriction (↓ renal blood flow), ↑ renin release, ↑ tubular Na⁺ reabsorption (stress/volume depletion response).
- RAAS: renin → angiotensin II (efferent constriction, ↑ proximal Na⁺ reabsorption) → aldosterone (ENaC ↑, K⁺ excretion ↑).
- Natriuretic peptides (ANP/BNP): promote natriuresis (↑ GFR, ↓ Na⁺ reabsorption) in volume overload states.
- ADH (vasopressin): water retention via aquaporin-2 insertion in collecting duct.
🧪 Core Physiology Themes (High-Yield)
| Theme 📌 | What’s happening? 🧠 | Clinical relevance 🩺 |
|---|---|---|
| Glomerular filtration (GFR) | Filtration depends on renal plasma flow and the balance of forces across the glomerular capillary. Afferent tone controls inflow; efferent tone controls outflow pressure and filtration fraction. | NSAIDs (afferent constriction) and ACEi/ARB (efferent dilation) can reduce GFR-especially in hypovolaemia, CKD, or renal artery stenosis. |
| Autoregulation 🎚️ | Kidneys stabilise GFR across a BP range using the myogenic response and tubuloglomerular feedback (macula densa sensing distal NaCl). | In shock/sepsis, autoregulation can fail → AKI risk rises. Careful fluid/blood pressure strategy matters. |
| Countercurrent system 🔁 | Juxtamedullary loops generate a corticomedullary osmotic gradient (multiplication) while vasa recta preserve it (exchange). ADH then “uses” this gradient to reclaim water in collecting ducts. | Early tubular injury often presents as impaired concentrating ability (polyuria, low urine osmolality) before creatinine rises. |
| Acid–base handling ⚗️ | PCT reabsorbs most filtered bicarbonate; distal nephron generates “new” bicarbonate and excretes acid (as NH₄⁺ and titratable acids). Intercalated cells are key. | Renal tubular acidosis patterns map to segment dysfunction (proximal vs distal) and have characteristic electrolyte profiles. |
📊 Cortex vs Medulla - Quick Comparison
| Feature | Cortex | Medulla |
|---|---|---|
| Main structures | Glomeruli, PCT, DCT | Loops of Henle, collecting ducts, vasa recta |
| Perfusion | High (majority of renal blood flow) | Low (physiologically low O₂) |
| Key job | Filtration + bulk reabsorption/secretion | Urine concentration (osmotic gradient) |
| Vulnerability | Perfusion-dependent; AKI in shock | Hypoxia prone; concentrating defects early |
🧠 Clinical Pearls (Renal Physiology → Bedside)
- 🩸 High flow, uneven oxygenation: cortex is well perfused; medulla is “on the edge” → hypoperfusion can cause tubular injury disproportionately.
- 💊 Drug effects map to anatomy: loop diuretics (TAL), thiazides (DCT), ACEi/ARB (efferent tone/RAAS), NSAIDs (afferent tone/prostaglandins).
- 🚑 Concentrating ability can fail early: tubular dysfunction may precede creatinine rise (watch urine output and osmolality when available).
- 🧪 Proteinuria is a glomerular clue: suggests filtration barrier injury-especially if persistent or heavy.
📝 Summary
Renal structure is tightly linked to renal function: glomeruli filter, PCT bulk-reabsorbs, the loop of Henle builds the medullary gradient, DCT fine-tunes electrolytes, and the collecting duct performs final water and acid–base control under ADH and aldosterone. The kidney’s unique “afferent → glomerulus → efferent” microcirculation supports filtration but creates segment-specific vulnerabilities-particularly medullary hypoxia and tubular injury in AKI.


Categories
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Critical care
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Geriatrics
- Guidelines
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Palliative Care
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Respiratory
- Resuscitation
- Revision Topics
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
