| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Philadelphia chromosome
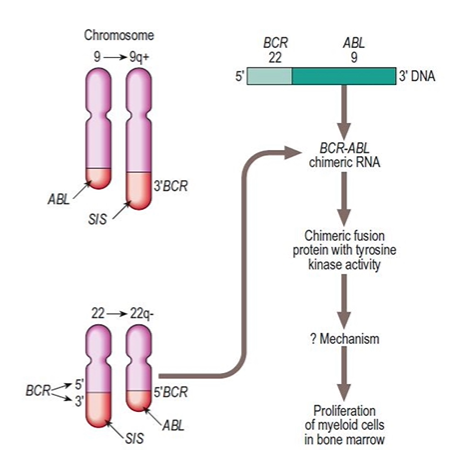
🧬 First discovered at the University of Pennsylvania by Peter Nowell in 1960, the Philadelphia chromosome was the first genetic abnormality directly linked to cancer. It is found in almost all cases of Chronic Myeloid Leukaemia (CML) and in some cases of Acute Lymphoblastic Leukaemia (ALL). This abnormality results from a reciprocal translocation between chromosomes 9 and 22 (t(9;22)), creating a shortened chromosome 22.
📖 About the Philadelphia Chromosome and CML
- Chromosomal Translocation: A segment of chromosome 9 swaps with chromosome 22, forming the Philadelphia chromosome (a shortened chromosome 22).
- BCR-ABL Fusion Gene: The ABL gene on chromosome 9 fuses with the BCR gene on chromosome 22 → producing a constitutively active tyrosine kinase. The main product in CML is p210, while p185 is often seen in Ph+ ALL.
- Pathophysiology: The BCR-ABL protein drives abnormal signalling → unchecked bone marrow proliferation, reduced apoptosis, and accumulation of myeloid cells.
- Targeted Therapy: 💊 Tyrosine kinase inhibitors (TKIs), e.g. imatinib, specifically block BCR-ABL activity, transforming CML from a fatal disease into a chronic, manageable condition.
- Abbreviations: BCR = breakpoint cluster region; ABL = Abelson tyrosine kinase.
⚡ Role of Tyrosine Kinases in Cell Biology
- Signalling Cascade: Tyrosine kinases mediate key cell signalling processes in growth, differentiation, metabolism, and apoptosis.
- Enzymatic Activity: They catalyse phosphorylation of tyrosine residues on target proteins → acting like “on/off switches” for cellular pathways.
- Oncogenesis: When dysregulated (e.g. BCR-ABL fusion), they become oncogenes driving malignancy.
- Impact of TKIs: Imatinib and newer agents (dasatinib, nilotinib, ponatinib) inhibit the aberrant kinase → halting proliferation and inducing remission.
🇬🇧 UK Clinical Pearls
- NICE recommends TKIs as first-line therapy for CML.
- Monitoring is via quantitative PCR for BCR-ABL transcripts (molecular response).
- Resistance mutations (e.g. T315I) may require switching to newer agents (ponatinib).
Categories
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Critical care
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Geriatrics
- Guidelines
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Palliative Care
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Respiratory
- Resuscitation
- Revision Topics
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
