| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Amino acids
Related Subjects: | Protein metabolism | Protein Synthesis | Amino acids | Human Metabolism
🧬 Amino Acids and Proteins in Medicine
🍖 Amino acids are the fundamental building blocks of proteins. Their side chains (R groups) determine charge, polarity, and reactivity, which in turn govern protein structure, enzyme catalysis, receptor binding, membrane transport, and many disease processes. For medical students, amino acid biochemistry is high-yield because it links directly to inborn errors of metabolism, nitrogen balance, liver failure/encephalopathy, nutrition, and protein misfolding disorders.
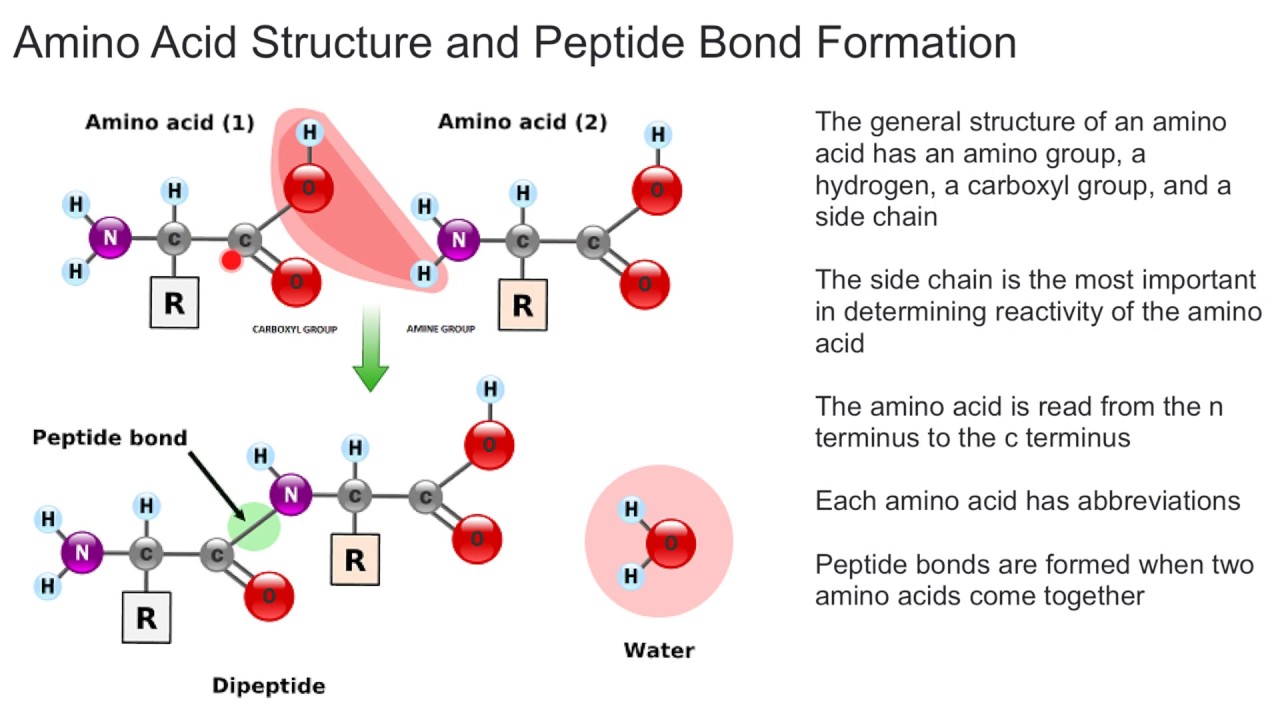
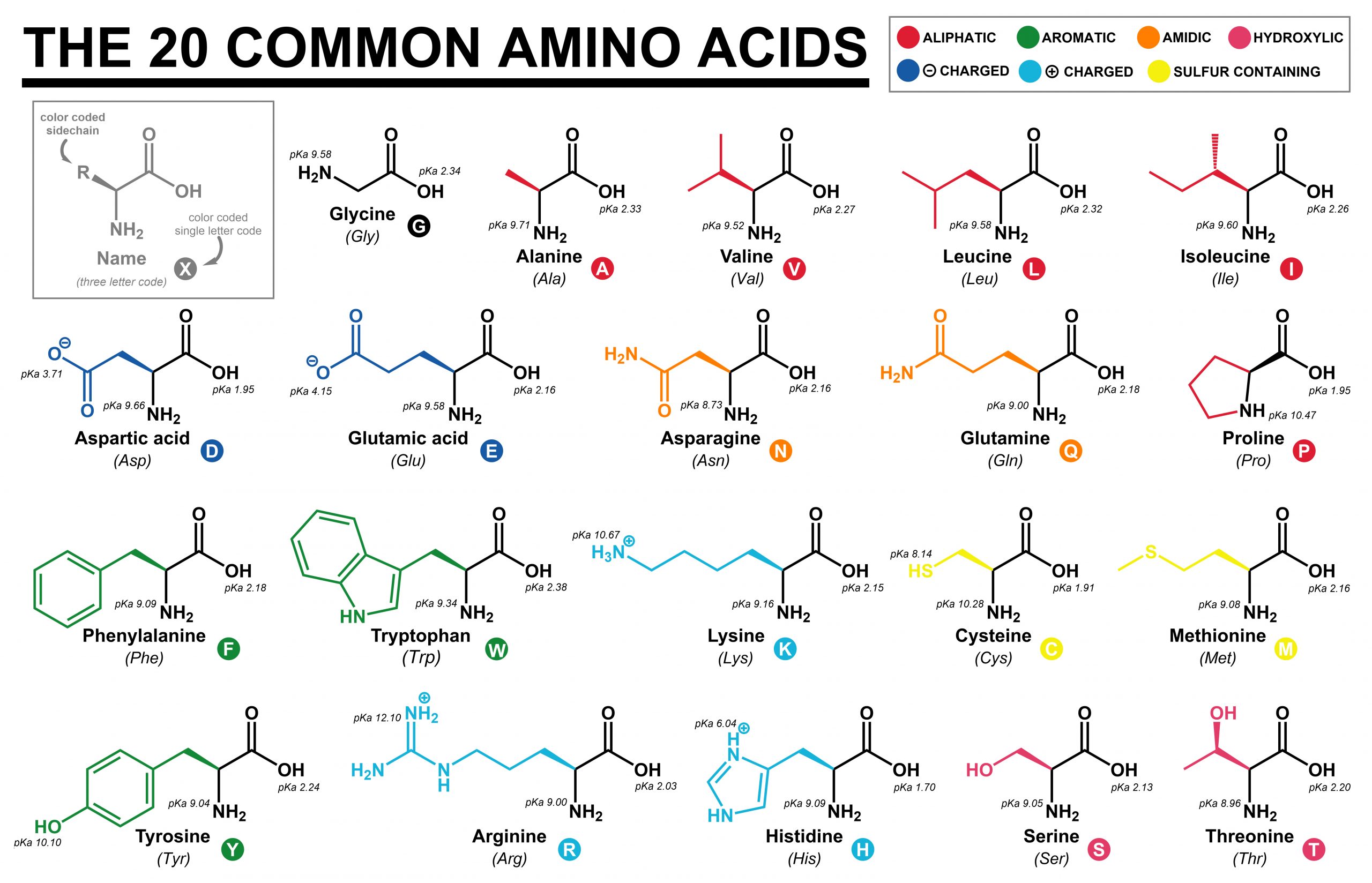
🔬 Amino Acid Structure: What Every Student Must Know
All standard amino acids incorporated into human proteins share the same core architecture:
- α-carbon (central carbon)
- α-amino group (–NH2, protonated to –NH3+ at physiological pH)
- α-carboxyl group (–COOH, deprotonated to –COO− at physiological pH)
- Hydrogen
- R group (side chain) - defines chemical behaviour
Zwitterions: At pH ~7.4, amino acids carry both positive and negative charges. This is why their charge changes with pH and why pKa and pI matter clinically (e.g., electrophoresis, protein binding, enzyme active sites).
🧩 Classifying Amino Acids (High-Yield)
1️⃣ By Polarity and Charge
- 🌑 Non-polar (hydrophobic): Gly, Ala, Val, Leu, Ile, Pro, Met, Phe, Trp
- 💧 Polar (uncharged): Ser, Thr, Cys, Tyr, Asn, Gln
- ⚡ Acidic (negative at pH 7.4): Asp, Glu
- ⚡ Basic (positive at pH 7.4): Lys, Arg, His
Pattern recognition: Hydrophobic residues tend to be buried inside globular proteins in aqueous environments, while polar/charged residues are surface-exposed. In membranes, the pattern reverses: hydrophobic residues face lipid.
2️⃣ By Nutritional Requirement
- 🥦 Essential (diet required): His, Ile, Leu, Lys, Met, Phe, Thr, Trp, Val
- 🍳 Non-essential: synthesised endogenously (many are formed by transamination)
- ⚠️ Conditionally essential: Arg, Cys, Gln, Gly, Pro, Tyr (important in illness, growth, stress, prematurity)
In exam terms: “conditionally essential” amino acids become limiting during catabolism, sepsis, major burns, critical illness, and childhood growth.
🧪 pKa, Isoelectric Point (pI), and Why They Matter
The pKa is the pH at which a group is 50% protonated and 50% deprotonated. Side-chain pKa values determine whether a residue is charged at physiological pH, influencing:
- Protein folding and stability
- Enzyme active-site chemistry
- Drug and ligand binding
- Buffering behaviour
- Histidine has an imidazole side chain with pKa ~6 → close to physiological pH → excellent buffer and common in enzyme active sites.
- Aspartate/Glutamate are negatively charged at pH 7.4 → contribute to binding of cations and positively charged ligands.
- Lysine/Arginine are positively charged → bind DNA/RNA (negatively charged phosphate backbone).
The isoelectric point (pI) is the pH at which the molecule has net zero charge. If pH > pI, net charge becomes negative; if pH < pI, net charge becomes positive. This is used in isoelectric focusing and 2D gel electrophoresis.
🧱 Proteins: Structure Levels and Clinical Correlates
1️⃣ Primary Structure
Linear amino acid sequence joined by peptide bonds (covalent amide links between –COO− and –NH3+). A single amino acid change can cause major disease (e.g., haemoglobinopathies).
2️⃣ Secondary Structure
- α-helix: stabilised by hydrogen bonds; 3.6 residues per turn. Proline introduces kinks and disrupts helices.
- β-sheet: parallel or antiparallel strands held by hydrogen bonds; common in structural proteins and amyloid.
3️⃣ Tertiary Structure
Overall 3D folding stabilised by:
- Hydrophobic interactions (major driver of folding in water)
- Hydrogen bonds
- Ionic bonds (salt bridges)
- Disulfide bonds (cysteine–cysteine, important in secreted proteins such as antibodies)
4️⃣ Quaternary Structure
Assembly of multiple polypeptide chains (e.g., haemoglobin). Quaternary interactions can underlie cooperativity and allosteric regulation.
Chaperones: Proteins often fold while being translated. Heat shock proteins and other chaperones prevent aggregation and misfolding - clinically relevant in stress states and neurodegeneration.
⚙️ Core Amino Acid Metabolism (Nitrogen + Carbon)
Amino acids are not stored as a dedicated pool. When dietary intake exceeds needs, amino acids are deaminated and their carbon skeletons are used for energy, glucose, ketones, or fat. Nitrogen must be safely handled to avoid toxicity.
🔁 Transamination (ALT/AST)
- Transfers an amino group to a keto acid to make a new amino acid
- Requires pyridoxal phosphate (vitamin B6)
- Clinically: ALT/AST rise in hepatocellular injury because these enzymes leak from damaged hepatocytes
➖ Oxidative Deamination
- Produces free ammonia (NH3) - toxic to the brain
- Ammonia is detoxified in the liver via the urea cycle
- Clinical relevance: liver failure → hyperammonaemia → encephalopathy
🧪 The Urea Cycle (Why It’s Clinically Dangerous When It Fails)
Urea cycle disorders lead to accumulation of ammonia, causing vomiting, lethargy, seizures, cerebral oedema, and coma, especially in neonates. Hyperammonaemia is a true emergency.
🔥 Glucogenic vs Ketogenic Amino Acids
- Glucogenic: carbon skeleton enters gluconeogenesis (most amino acids)
- Ketogenic: carbon skeleton forms ketone bodies (classically Leucine and Lysine)
- Both: some contribute to both pathways (e.g., isoleucine, phenylalanine, tyrosine, tryptophan)
High-yield clinical link: during fasting, muscle amino acids support hepatic gluconeogenesis (notably via the alanine cycle) - relevant in cachexia, sepsis, and diabetes.
🧠 Amino Acids as Precursors (Very Exam-Friendly)
- Tryptophan → serotonin, melatonin, niacin
- Tyrosine → dopamine, noradrenaline, adrenaline; also thyroid hormones (T3/T4)
- Histidine → histamine
- Glutamate → GABA
- Arginine → nitric oxide (vasodilation signalling)
- Glycine → part of haem synthesis; inhibitory neurotransmitter in spinal cord
🏥 Clinical Syndromes and Inborn Errors of Amino Acid Metabolism
1️⃣ Phenylketonuria (PKU)
- Defect: phenylalanine hydroxylase (or BH4 cofactor deficiency)
- Biochemistry: ↑ phenylalanine → neurotoxicity; ↓ tyrosine (tyrosine becomes “essential”)
- Clinical: intellectual disability if untreated, seizures, eczema-like rash, “musty” odour
- Management: dietary phenylalanine restriction; BH4 (sapropterin) in selected cases
2️⃣ Maple Syrup Urine Disease (MSUD)
- Defect: branched-chain α-ketoacid dehydrogenase
- Amino acids: ↑ Leu, Ile, Val (branched-chain AAs)
- Clinical: poor feeding, lethargy, seizures, encephalopathy; maple syrup odour
- Management: dietary restriction; emergency management during decompensation
3️⃣ Homocystinuria
- Defect: commonly cystathionine β-synthase (B6-related pathway)
- Biochemistry: ↑ homocysteine and methionine
- Clinical: early vascular disease/thrombosis, lens dislocation, marfanoid habitus
- Management: B6, folate/B12 support, betaine, dietary methionine restriction (specialist)
4️⃣ Urea Cycle Disorders
- Core problem: ammonia detoxification fails → hyperammonaemic encephalopathy
- Clues: neonate with vomiting, lethargy, respiratory alkalosis, seizures
- Management principle: reduce ammonia urgently and stop protein temporarily under specialist care
🥗 Protein-Energy Malnutrition (PEM)
- Kwashiorkor: relative protein deficiency → oedema (↓ albumin), fatty liver, skin/hair changes, infections
- Marasmus: severe calorie deficiency → marked muscle wasting, loss of subcutaneous fat, “skin and bone”
In hospital medicine, protein deficiency presents as poor wound healing, sarcopenia, immune dysfunction, pressure sores, and frailty syndromes. Nutrition is a major determinant of outcomes after surgery, infection, and critical illness.
🧠 Protein Misfolding and Deposition Disorders
Protein misfolding can produce insoluble aggregates that disrupt tissues:
- Amyloidosis: extracellular deposition of misfolded proteins (β-sheet rich), leading to organ dysfunction
- Alzheimer disease: amyloid-β plaques and tau neurofibrillary tangles
- Prion diseases (e.g., CJD): infectious protein conformational change (α-helix → β-sheet), protease-resistant aggregates
- Heinz bodies: denatured haemoglobin aggregates on RBC membrane (e.g., oxidative stress, G6PD deficiency)
Medical reasoning link: “Protein structure” is not abstract - misfolding explains real disease mechanisms in neurology, haematology, and systemic amyloidosis.
🩸 Albumin and Plasma Proteins (Clinical Relevance)
- Albumin: maintains oncotic pressure; binds many drugs and hormones; low in inflammation (negative acute-phase reactant)
- Immunoglobulins: antibody proteins (structure relies on disulfide bonds)
- Clotting factors: proteins with vitamin K–dependent γ-carboxylation (clinically relevant to warfarin)
Hypoalbuminaemia usually reflects inflammation, capillary leak, malnutrition, liver disease, or nephrotic loss. It is a prognostic marker and affects drug binding and oedema risk.
💊 Therapeutic and Practical Applications
- TPN: amino-acid mixtures provide nitrogen when gut feeding not possible
- Enteral feeds: high-protein formulas in catabolism, critical illness, burns
- Renal disease: protein restriction may be used selectively; dialysis increases amino acid losses
- Liver disease: nutrition is essential; some use branched-chain amino acids in selected settings
- Metabolic disorders: targeted amino acid restriction (e.g., PKU, MSUD)
🧠 Exam Pearls (Quick High-Yield)
- Histidine buffers near physiological pH → active sites and Hb function
- Proline disrupts α-helices; important in collagen structure
- Cysteine forms disulfide bonds; redox-sensitive
- Leucine + Lysine are purely ketogenic
- ALT/AST are transaminases (B6-dependent) → key liver injury markers
- Hyperammonaemia = emergency (urea cycle failure or severe liver dysfunction)
📌 Summary
Amino acids are chemically diverse molecules whose side chains determine protein folding, enzyme catalysis, signalling pathways, and binding interactions. Proteins operate at every level of physiology, and disruption of amino acid handling or protein structure leads to clinically important disease: inborn errors (PKU, MSUD, homocystinuria), hyperammonaemia, malnutrition syndromes, and misfolding disorders (amyloid, prions). Mastering amino acid chemistry provides a foundation for understanding metabolism, nutrition, neurology, liver disease, and systemic pathology.
Categories
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
